by Christopher L. Grachanen
Calibration and Testing laboratories seeking accreditation status or maintaining their existing accreditation status must comply with the requirements denoted in ISO/IEC 17025 General requirements for the competence of testing and calibration laboratories, current edition 2017-11.
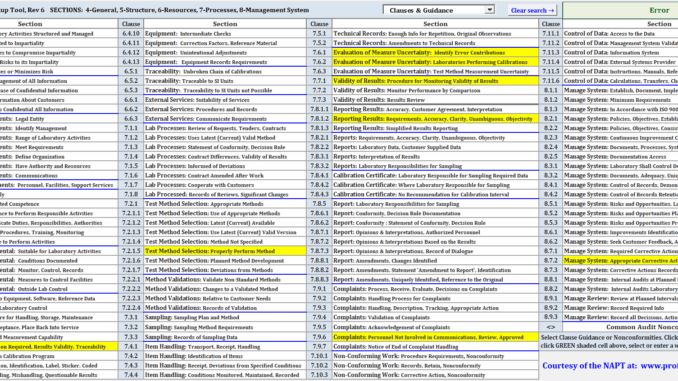
ISO/IEC 17025 requirements, identified by clause numbers, give details as to activities, policies, procedures and documentation necessities that laboratories need be able to demonstrate satisfactory fulfillment of. The ISO/IEC 17025 standard is +30 pages, comprised of five main sections that include Section 4 (General Requirements), Section 5 (Structural Requirements), Section 6 (Resource Requirements), Section 7 (Process Requirements), and Section 8 (Management Systems Requirements). The standard has twenty four clause groupings and 147 specific clause requirements. The first three clauses cover scope, normative references, and terms and definitions.
Assessors and laboratory personnel tasked in accreditation compliance must not only be familiar with these clauses but also the interdependencies between clauses which typically requires flipping through paper pages or searching through an electronic edition of the standard.
The vast majority of these clauses do not provide guidance or examples for clause fulfillment often require referencing other publications to determine compliance, which can be both daunting and time consuming. Additionally, example nonconformances related to a clause are also not provided which would otherwise provide valuable insight on things to be avoided. It was during an A2LA assessor class final exam that the author realized the need of a more efficient means to access ISO/IEC 17025 clauses with additional guidance for individuals not intimately familiar with the standard and clauses minutia.



