Articles
Accreditation to ISO/IEC 17025: Understanding the Benefits and the Process
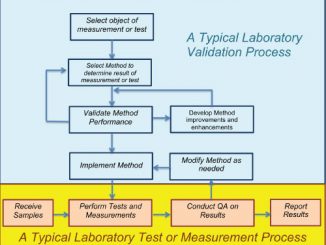
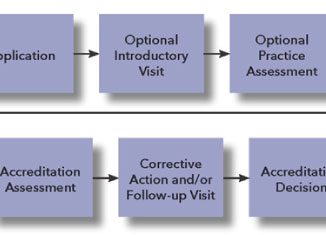
by Roger Muse To the uninitiated, accreditation to ISO/IEC 17025 can seem mysterious, confusing, or downright daunting. Just what does accreditation mean? And exactly how is it accomplished? >> Read Full Article (PDF)